Latest Updates
-
 Happy Birthday Arijit Singh: Start Your Weekend With 7 Songs That Match Every Mood From Morning To Midnight
Happy Birthday Arijit Singh: Start Your Weekend With 7 Songs That Match Every Mood From Morning To Midnight -
 The Creamy Side Dish Trick: Perfect Mashed Potato Recipe
The Creamy Side Dish Trick: Perfect Mashed Potato Recipe -
 Uranus In Gemini Transit Returns For The First Time Since The 1940s, Starts A 7-Year Shift Across Zodiac Signs
Uranus In Gemini Transit Returns For The First Time Since The 1940s, Starts A 7-Year Shift Across Zodiac Signs -
 Sita Navami 2026: Puja Muhurat, Vrat Vidhi And Spiritual Benefits For Stronger Relationships
Sita Navami 2026: Puja Muhurat, Vrat Vidhi And Spiritual Benefits For Stronger Relationships -
 World Malaria Day 2026: Date, History, Significance, and Why It Matters
World Malaria Day 2026: Date, History, Significance, and Why It Matters -
 Bakery Style Soft Texture Banana Cake Recipe
Bakery Style Soft Texture Banana Cake Recipe -
 Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains
Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains -
 Rich Mughlai Special Chicken Korma Recipe
Rich Mughlai Special Chicken Korma Recipe -
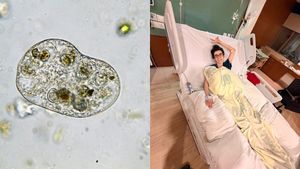 A Hidden Foodborne Infection: What You Should Know About Cyclospora
A Hidden Foodborne Infection: What You Should Know About Cyclospora -
 Melt-in-Mouth Sweet Mysore Pak Recipe: A Classic Indian Delight
Melt-in-Mouth Sweet Mysore Pak Recipe: A Classic Indian Delight
Serum Institute CEO Hails WHO's Approval Of Covovax For Emergency Use
Serum Institute of India (SII) CEO Adar Poonawalla on Friday hailed WHO's approval for emergency use of the Covovax vaccine "as yet another milestone" in the fight against Covid-19.
The World Health Organization (WHO) on Friday listed the Covovax as the ninth Covid-19 vaccine for emergency use with an aim to increase access to vaccination in lower-income countries.
It is produced by the Serum Institute of India under the licence of Novavax.
Reacting to the development, Poonawalla tweeted, "This is yet another milestone in our fight against Covid-19, Covovax is now WHO approved for emergency use, showing excellent safety and efficacy. Thank you all for a great collaboration..."
He tagged Novavax, WHO, Gavi, the Vaccine Alliance, its CEO Seth Berkley and the Gates Foundation. Currently, Covishield and other Covid-19 vaccines are approved for people above the age of 18.
According to WHO, Covovax was assessed under its emergency use listing (EUL) procedure based on the review of data on quality, safety and efficacy, a risk management plan, programmatic suitability, and manufacturing site inspections carried out by the Drugs Controller General of India.
"The Technical Advisory Group for Emergency Use Listing (TAG-EUL), convened by WHO and made up of experts from around the world, has determined that the vaccine meets WHO standards for protection against Covid-19, that the benefit of the vaccine far outweighs any risks, and that the vaccine can be used globally," it said.
Disclaimer: The information provided in this article is for general informational and educational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or a qualified healthcare provider with any questions you may have regarding a medical condition.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications
