Latest Updates
-
 Happy Birthday Arijit Singh: Start Your Weekend With 7 Songs That Match Every Mood From Morning To Midnight
Happy Birthday Arijit Singh: Start Your Weekend With 7 Songs That Match Every Mood From Morning To Midnight -
 The Creamy Side Dish Trick: Perfect Mashed Potato Recipe
The Creamy Side Dish Trick: Perfect Mashed Potato Recipe -
 Uranus In Gemini Transit Returns For The First Time Since The 1940s, Starts A 7-Year Shift Across Zodiac Signs
Uranus In Gemini Transit Returns For The First Time Since The 1940s, Starts A 7-Year Shift Across Zodiac Signs -
 Sita Navami 2026: Puja Muhurat, Vrat Vidhi And Spiritual Benefits For Stronger Relationships
Sita Navami 2026: Puja Muhurat, Vrat Vidhi And Spiritual Benefits For Stronger Relationships -
 World Malaria Day 2026: Date, History, Significance, and Why It Matters
World Malaria Day 2026: Date, History, Significance, and Why It Matters -
 Bakery Style Soft Texture Banana Cake Recipe
Bakery Style Soft Texture Banana Cake Recipe -
 Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains
Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains -
 Rich Mughlai Special Chicken Korma Recipe
Rich Mughlai Special Chicken Korma Recipe -
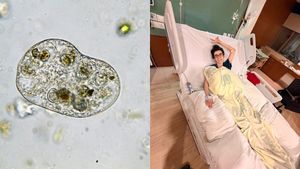 A Hidden Foodborne Infection: What You Should Know About Cyclospora
A Hidden Foodborne Infection: What You Should Know About Cyclospora -
 Melt-in-Mouth Sweet Mysore Pak Recipe: A Classic Indian Delight
Melt-in-Mouth Sweet Mysore Pak Recipe: A Classic Indian Delight
COVID-19 Vaccine India Registration, Eligibility: 22 Frequently Asked Questions (FAQs) Addressed
With the approval of two COVID-19 vaccines (Oxford's Covishield and Bharat Biotech's Covaxin) by the Drug Controller General of India (DCGI), the head of the Indian drug regulatory body, the nation is all set to combat the coronavirus which has affected around 85.7 million people and caused 1.85 million deaths worldwide (as of 5 January, 2:47 pm).

In this regard, the Ministry of Health & Family Welfare (Mohfw) has released a set of frequently asked questions or faqs for the general public to give them a better understanding of the vaccination process, especially related to its safety, efficacy and doses.
In this article, we will discuss all the questions related to COVID-19 vaccine in India which are answered by the Mohfw. Take a look.
1. When will India launch its vaccination drive against the COVID-19?
As per the announcement by the government followed by Mr Modi's tweet, the two vaccines named Covishield and Covaxin is all set to rollout on 16 January, with an aim to vaccinate around 300 million people by July.
2. Will COVID-19 vaccine be given to everyone simultaneously?
No, as per the government, people will be divided into groups and those who are at high-risk groups will be vaccinated on priority. The high-risk group or people in the first group will include healthcare and frontline workers. The second group will include people who are above 50 years and those with pre-existing medical conditions. Subsequently, the vaccine will be given to all in need.
3. Will the vaccine be safe as it has been being introduced in a short span of time?
Vaccines will be introduced in the country only after getting approved by the Drug Controller General of India (DCGI), the head of the Indian drug regulatory body. They will test the vaccine based on its safety and efficacy and will then introduce it for the public.
4. Is it mandatory to take the vaccine?
Vaccines not only help the immune system to make antibodies for a particular virus, but also help provide a broad immune system response. Though the vaccination for COVID-19 is voluntary which means healthy adults can take it as per their wish, it is advised to everyone to take the complete dose of the vaccine to protect oneself from COVID-19 and limit the spread of the virus.
5. Is it necessary for a COVID-19 recovered individual to take the vaccine?
According to many recent published studies, people who have recovered from COVID-19 have potent infection-blocking antibodies against the virus. However, it is advisable to receive the complete dose of the vaccine irrespective of the past history of infection. This is because the COVID-19 vaccine will not only protect us from the current strain of the virus, but will also help boost the immune system response from all aspects. COVID-19 vaccine can also be effective for its new strain, however, more research is needed in the area as no such statements are made on the effectiveness of the vaccine against the new coronavirus strain.
6. Can a person with COVID-19 (confirmed/suspected) be vaccinated?
People who are COVID-19 positive or are on the list of suspected cases are advised to postpone the vaccination for around 14 days. This will help them evaluate their symptoms and prevent the risk of virus spread at the site of vaccination.
7. Out of the multiple vaccines available, how is one or more vaccines chosen for administration?
The DCGI will give licence to vaccines only after evaluating their safety and efficacy based on data from clinical trials. As of now, the government of India has approved only two vaccines which are Oxford's Covishield and Bharat Biotech's Covaxin. The only thing to ensure is out of these two approved vaccines, one needs to complete the dose by only one type of vaccine, as both of them can have different rates of safety and effectiveness.
8. At what temperature do the vaccines need to be stored?
According to experts, both the vaccines can be stored at temperatures between 2°C and 8°C.
9. Does India have the facility to store and transport the COVID-19 vaccines at that temperature?
India runs one of the largest immunisation programmes in the world, catering the vaccination needs of over 26 million newborns and 29 million pregnant women. Also, for the development of Oxford's Covishield, the Serum Institute of India is their manufacturing and trial partner. This shows that India's vaccination program is strong and effective to cater to the needs of the country's large and diverse population.

10. Will the vaccines approved in India be as effective as those introduced in other countries?
Yes, the COVID-19 vaccines introduced in India can be as effective as vaccines developed by other countries. This is because many phases of clinical trials are carried out to ensure the safety and efficacy of all those vaccines. `
11. How to know the eligibility for vaccination?
Eligible individuals will be informed about the vaccination and its scheduled time through their registered mobile number.
12. What documents are required for registration by the eligible individuals?
Eligible individuals are required to bring any of the below mentioned photo IDs at the time of registration.
- Aadhaar/Driving License/Voter ID/PAN Card/Passport/ Job Card/Pension Document
- Health Insurance Smart Card issued under the scheme of Ministry of Labour
- Mahatma Gandhi National Rural Employment Guarantee Act (MGNREGA)
- Official identity cards issued to MPs/MLAs/MLCs
- Passbooks issued by Bank/Post Office
- Service ID Card issued by Central/State Govt:/Public Limited Companies
13. Can a person get the COVID-19 vaccine without registration?
No, registration prior to the vaccine shot is mandatory as information of the session site and time will be shared to the individual through their registered mobile number.
14. If a person is unable to bring the required photo ID at the site of vaccination, will he/she be allowed for the process?
Photo ID is required for both registration and verification at the site of the vaccination to ensure that the intended person is vaccinated.

15. How will the beneficiary receive the information about their vaccination due date?
After online registration, the individual will receive details via SMS on their registered mobile number regarding the date, place and time of vaccination.
16. Will beneficiaries receive information about their vaccination status on completion?
Yes, after getting the vaccine shot, the individual will be notified by a message on their registered mobile number. Details of the second dose will also be shared by the individual. When all the vaccine doses will be completed, the person will receive a QR code-based certificate on their registered mobile number.
17. Are there any precautionary or preventive measures to follow at the site or place of vaccination?
The precautionary or preventive measures to be followed by each individual are:
- After the administration of the vaccine, the candidate must rest at the vaccination centre for at least half an hour to check for any noticeable side effects such as headache, nausea, etc.
- If any such side effects, discomfort or uneasiness persists, the nearest health authorities/ANM/ASHA must be informed immediately.
- At the site of vaccination, all the existing norms must be followed such as wearing the mask, hand hygiene and physical distancing.
18. Are there any possible side effects of COVID-19 vaccine?
As of now, there's no such information available regarding the possible side effects of COVID-19 vaccine. However, like other vaccine shots, some of the common side effects are possible such as mild fever, headache, pain in the site of injection, nausea and many others. Also, states have been asked to make necessary arrangements to deal with any such COVID-19 vaccine-related side effects.
19. If a person is on medications for his/her pre-existing conditions such as diabetes, blood pressure or cancer, can they take COVID-19 vaccine?
Yes, as aforementioned, people with any underlying or pre-existing medical conditions are in the second high-risk group of people, with healthcare and frontline workers being in the first group. Considering the ongoing medications of these people, their vaccine dose may be altered by medical experts.
20. Will the family of healthcare providers or frontline workers be also given the vaccine?
At the initial phase, only people in the high-risk group will be given vaccination, followed by the second high-risk group of people. Later, it will be made available to all in need.
21. How many doses of the vaccine are available and what will be the gap between the doses?
According to the reports, both Covishield and Covaxin have two full doses. After the registration of the first dose, the second dose will be given after 28 days.
22. When will the antibodies start developing after the dose?
According to clinical trial data of both the approved vaccines, the antibodies will generally start developing two weeks after receiving the second dose of COVID-19 vaccine. This is why, it is advised to not miss the second dose as only after a full vaccine dose, the combined analysis of its efficacy can be made.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications
