Latest Updates
-
 If You Have PCOS, Summer Might Be Affecting You More Than You Think
If You Have PCOS, Summer Might Be Affecting You More Than You Think -
 Akshaya Tritiya 2026: What to Buy and Avoid for Good Luck, According to Rinhee Suberwal
Akshaya Tritiya 2026: What to Buy and Avoid for Good Luck, According to Rinhee Suberwal -
 Akshaya Tritiya 2026: 6 Trendy Gold Jewellery Designs You’ll Wear Beyond The Festive Day
Akshaya Tritiya 2026: 6 Trendy Gold Jewellery Designs You’ll Wear Beyond The Festive Day -
 Do Men Who Listen to Their Wives Succeed More? What Research Really Says
Do Men Who Listen to Their Wives Succeed More? What Research Really Says -
 Dal Palak Recipe: Your Healthy Green Dal Twist
Dal Palak Recipe: Your Healthy Green Dal Twist -
 Tremors in Kashmir After Afghanistan Quake: What Residents Experienced and Precautions to Take
Tremors in Kashmir After Afghanistan Quake: What Residents Experienced and Precautions to Take -
 Who Is Shaheen Bhatt’s Fiancé Ishaan Mehra? Inside the Engagement That Left Alia Bhatt ‘Weeping’
Who Is Shaheen Bhatt’s Fiancé Ishaan Mehra? Inside the Engagement That Left Alia Bhatt ‘Weeping’ -
 World Heritage Day 2026: Significance, Theme And The Cost Of Failing To Protect Our Past
World Heritage Day 2026: Significance, Theme And The Cost Of Failing To Protect Our Past -
 South Style Tangy Side: The Ultimate Tomato Chutney Recipe
South Style Tangy Side: The Ultimate Tomato Chutney Recipe -
 Horoscope for Today April 18, 2026 - Steady Progress, Calm Momentum
Horoscope for Today April 18, 2026 - Steady Progress, Calm Momentum
EU Regulator Approves BioNTech-Pfizer Vaccine For Children In 5-11 Age Group
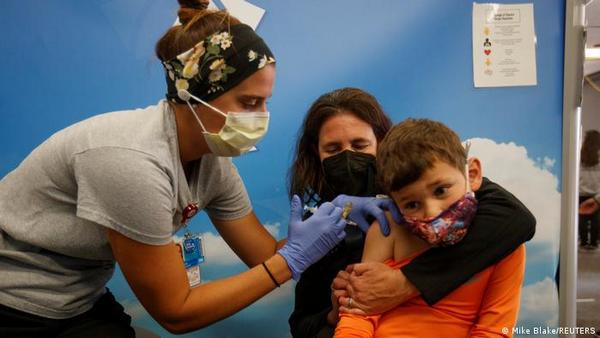
The European Medicines Agency, the EU's regulator for medicinal products, on Thursday approved the BioNTech-Pfizer vaccine, also known as Comirnaty, for children aged 5-11. It's the first vaccine approved for children under 12 in the 27-member EU.The vaccine has already been approved in the EU for use in people between the ages of 12 and 17 years since May and for adults since December 2020.
EMA said the vaccine should be given to children in two doses of 10 micrograms three weeks apart as an injection in the upper arm. Adult doses contain 30 micrograms. "The benefits of Comirnaty in children aged 5 to 11 outweigh the risks, particularly in those with conditions that increase the risk of severe COVID-19," EMA said.
Vaccinating children seen as key
The companies said a clinical trial of children aged 5 to 11 showed their vaccine to be 90.7% effective against the coronavirus. However, the studies have not been large enough to determine whether the vaccine could produce rare side effects such as chest and heart inflammation that have been seen in mostly male older teenagers and young adults after the second dose.
The approval for younger children comes as Europe battles another massive surge of COVID-19 cases. Authorities in Austria, which is seeing a very high rate of infections, did not wait for EMA approval and have already begun vaccinating the 5 to 11 age group.
In Germany and the Netherlands, children now account for the majority of cases, and experts say that vaccinating them could be vital to bringing the fourth wave of the pandemic under control. COVID-19 death in Germany reached 100,000 on Thursday.
What happens now?
EMA approval now has to be rubber-stamped by the EU's executive branch, the European Commission, before the vaccination can be rolled out for the 5-11 age group in EU member states. It typically follows EMA guidelines.
German Health Minister Jens Spahn said earlier this week that shipping of vaccines for younger children in the EU would begin on December 20. Germany's standing vaccination committee (STIKO) is due to issue its recommendation on vaccinating children aged 5-11 before the end of the year.
tj/msh (Reuters, AFP)



 Click it and Unblock the Notifications
Click it and Unblock the Notifications
