Latest Updates
-
 Restaurant Style Paneer Masala Recipe for a Perfect Dinner
Restaurant Style Paneer Masala Recipe for a Perfect Dinner -
 Bagalamukhi Jayanti 2026 Date: When ‘Dhurandhar’ Fame Aditya Dhar Visited Bagalamukhi Temple With Yami Gautam
Bagalamukhi Jayanti 2026 Date: When ‘Dhurandhar’ Fame Aditya Dhar Visited Bagalamukhi Temple With Yami Gautam -
 Ritesh Bawri’s Journey from 14 Near-Death Experiences to Reversing Chronic Illness
Ritesh Bawri’s Journey from 14 Near-Death Experiences to Reversing Chronic Illness -
 Crispy South Indian Snack: The Ultimate Medu Vada Recipe
Crispy South Indian Snack: The Ultimate Medu Vada Recipe -
 Who Is Sadhvi Satish Sail? The Journey From Goa To Miss India World 2026 Crown
Who Is Sadhvi Satish Sail? The Journey From Goa To Miss India World 2026 Crown -
 Makeup Hacks 101: How to Keep Your Makeup Fresh in Summer Without Overdoing It
Makeup Hacks 101: How to Keep Your Makeup Fresh in Summer Without Overdoing It -
 Maharashtra Restaurants Must Declare Fake Paneer From May 1: How To Spot Fake Vs Real Paneer
Maharashtra Restaurants Must Declare Fake Paneer From May 1: How To Spot Fake Vs Real Paneer -
 Asthma, Medication, and Weight: Why Anant Ambani May Not Be Losing Weight Easily
Asthma, Medication, and Weight: Why Anant Ambani May Not Be Losing Weight Easily -
 Celeb Beauty Routine: Inside Bigg Boss 17 Fame Soniya Bansal’s Everyday Skincare Ritual
Celeb Beauty Routine: Inside Bigg Boss 17 Fame Soniya Bansal’s Everyday Skincare Ritual -
 Light Healthy Meal: The Ultimate Vegetable Soup Recipe
Light Healthy Meal: The Ultimate Vegetable Soup Recipe
Covid-19 Vaccines For Kids: DCGI Grants Emergency Use Nod To 3 Vaccines For Different Age Group Of Children
On Tuesday (26 April), the Drugs Controller General of India (DCGI) approved three Covid-19 vaccines for restricted emergency use authorisation (EUA) in children of various ages.
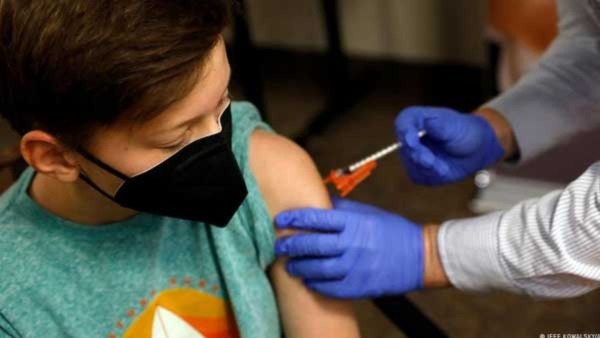
Bharat Biotech's Covaxin received a restricted EUA from the DCGI for children aged 6 to12. Similarly, Biological E's Covid-19 vaccine Corbevax has been approved for children aged 5 to 12 years.
Further, along with Corbevax and Covaxin, the DCGI has also approved Zydus Cadila's Covid-19 vaccine ZyCoV-D for children over the age of 12 years for a two-dose regimen.
This development happened after the Subject Expert Committee's (SEC) discussion of suggestions for the emergency use of Covaxin in children aged 6 to 12 years due to an increase in covid-19 cases in schools.
Covaxin has proven to be effective for adults and is also presently being given to youngsters aged 15 to 18. To give EUA to Covaxin for children aged 6-12 years, experts had requested more information from the corporation, which was delivered on Friday. The SEC was satisfied with the provided data and thus, nods for its EUA among children aged 6-12 years.
As the focus was mainly on the increasing number of cases among children, the SEC has also recommended emergency use of Biological E's Covid-19 vaccine Corbevax and ZyCoV-D.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications
