Latest Updates
-
 Rihanna Brings Indian Twist Wearing ‘Haathphool’ At Fenty Night In Mumbai, Isha Ambani Stuns In Dior Couture
Rihanna Brings Indian Twist Wearing ‘Haathphool’ At Fenty Night In Mumbai, Isha Ambani Stuns In Dior Couture -
 Happy Birthday Arijit Singh: Start Your Weekend With 7 Songs That Match Every Mood From Morning To Midnight
Happy Birthday Arijit Singh: Start Your Weekend With 7 Songs That Match Every Mood From Morning To Midnight -
 The Creamy Side Dish Trick: Perfect Mashed Potato Recipe
The Creamy Side Dish Trick: Perfect Mashed Potato Recipe -
 Uranus In Gemini Transit Returns For The First Time Since The 1940s, Starts A 7-Year Shift Across Zodiac Signs
Uranus In Gemini Transit Returns For The First Time Since The 1940s, Starts A 7-Year Shift Across Zodiac Signs -
 Sita Navami 2026: Puja Muhurat, Vrat Vidhi And Spiritual Benefits For Stronger Relationships
Sita Navami 2026: Puja Muhurat, Vrat Vidhi And Spiritual Benefits For Stronger Relationships -
 World Malaria Day 2026: Date, History, Significance, and Why It Matters
World Malaria Day 2026: Date, History, Significance, and Why It Matters -
 Bakery Style Soft Texture Banana Cake Recipe
Bakery Style Soft Texture Banana Cake Recipe -
 Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains
Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains -
 Rich Mughlai Special Chicken Korma Recipe
Rich Mughlai Special Chicken Korma Recipe -
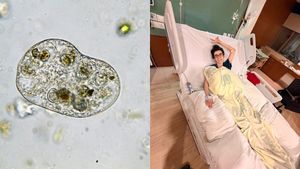 A Hidden Foodborne Infection: What You Should Know About Cyclospora
A Hidden Foodborne Infection: What You Should Know About Cyclospora
Phase III Trials Of Covid Drug Movfor Show Positive Results, Says Hetero
Drug firm Hetero on Saturday announced positive results of Phase III trials of Movfor (Molnupiravir), an investigational oral antiviral COVID-19 medicine.
The results, presented as an Oral Abstract at the Conference on Retroviruses and Opportunistic Infections (CROI) held virtually February 12-16, 2022, demonstrated that Molnupiravir along with the standard of care (SOC) reduced the risk of hospitalisation by over 65 per cent compared to SOC alone.

Early viral clearance (negative RT-PCR) and significant clinical improvement were observed within five days of administering the antiviral drug, the Hyderabad-based drugmaker said in a statement.
No fatalities were reported during the study, it added.
Under the Phase III trials, one of the studies included 1,218 COVID-19 patients, it said. The study enrolled eligible patients, within five days of symptom onset, were administered with Molnupiravir capsules(800 mg twice daily) for five days, it added.
Hetero entered into a non-exclusive voluntary licensing agreement with MSD in April 2021 for the manufacturing and distribution of Molnupiravir for the treatment of COVID-19.
Under this licensing deal, Hetero was allowed to expand access to Molnupiravir in India and in other low-and middle-income countries (LMICs), following the approvals for emergency use authorisation by local regulatory agencies.
Disclaimer: The information provided in this article is for general informational and educational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or a qualified healthcare provider with any questions you may have regarding a medical condition.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications
