Latest Updates
-
 Sita Navami 2026: Puja Muhurat, Vrat Vidhi And Spiritual Benefits For Stronger Relationships
Sita Navami 2026: Puja Muhurat, Vrat Vidhi And Spiritual Benefits For Stronger Relationships -
 World Malaria Day 2026: Date, History, Significance, and Why It Matters
World Malaria Day 2026: Date, History, Significance, and Why It Matters -
 Bakery Style Soft Texture Banana Cake Recipe
Bakery Style Soft Texture Banana Cake Recipe -
 Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains
Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains -
 Rich Mughlai Special Chicken Korma Recipe
Rich Mughlai Special Chicken Korma Recipe -
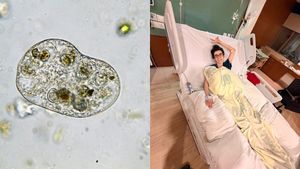 A Hidden Foodborne Infection: What You Should Know About Cyclospora
A Hidden Foodborne Infection: What You Should Know About Cyclospora -
 Melt-in-Mouth Sweet Mysore Pak Recipe: A Classic Indian Delight
Melt-in-Mouth Sweet Mysore Pak Recipe: A Classic Indian Delight -
 Between Meetings and Meals: Why American Pecans Are the Ideal Midday Snack
Between Meetings and Meals: Why American Pecans Are the Ideal Midday Snack -
 Skincare Hacks 101: 7 Summer Hacks That Actually Work in 40°C Heat
Skincare Hacks 101: 7 Summer Hacks That Actually Work in 40°C Heat -
 8 Workouts That Should Be a Part of Everyone’s Lifestyle for Longevity
8 Workouts That Should Be a Part of Everyone’s Lifestyle for Longevity
Johnson & Johnson Gets Nod For Emergency Use: Everything You Need To Know About This Single-Dose Vaccine
What You Need To Know About J&J Covid-19 Vaccine
Credit: Reuters
As part of expanding its vaccine basket, India has given Emergency Use Authorisation (EUA) to the single-dose vaccine developed by Janssen Pharmaceuticals, a Belgium based subsidiary of the global major Johnson & Johnson.
Johnson & Johnson is the fifth vaccine to get approval for emergency use, after Covishield by AstraZeneca, Covaxin by Bharat Biotech Sputnik V by Russia's Gamaleya Institute and Moderna by Moderna Inc.
The US drugmaker Johnson & Johnson applied for Emergency Use Authorisation of their COVID-19 vaccine in India on August 5. The company claims the vaccine has proved to be 85 per cent effective in the clinical trials; the vaccine has also demonstrated resistance against Delta and other emerging variants. Hyderabad-based vaccine maker Biological E Ltd will produce the vaccine in the country.
Johnson & Johnson vaccine was issued an emergency use authorisation in the United States by the Food and Drug Administration on February 27, 2021. The European Union authorized the vaccine for emergency use as well.
"Johnson and Johnson's single-dose Covid-19 vaccine is given approval for Emergency Use in India. Now India has 5 EUA vaccines. This will further boost our nation's collective fight against COVID-19," Union Health Minister Mansukh Mandaviya tweeted on Sunday, 8 August 2021 [1].
"We are pleased to announce that on August 7, 2021, the Government of India issued Emergency Use Authorisation (EUA) for the Johnson & Johnson Covid-19 single-dose vaccine in India, to prevent Covid-19 in individuals 18 years of age and older," Johnson & Johnson's India spokesperson said in a statement as quoted by news agency ANI [2].
How Johnson & Johnson vaccine works
The single-dose vaccine, known as JNJ-78436735 or Ad26.COV2.S. was developed by Janssen Pharmaceutica, a Belgium-based division of Johnson & Johnson, in collaboration with Beth Israel Deaconess Medical Center.
Unlike Moderna and Pfizer which use mRNA technology-messenger RNA is a code that instructs the immune system to recognise and attack the COVID-19 virus- Johnson and Johnson's viral vector vaccine uses a disabled adenovirus.
The adenovirus is not linked to coronavirus and is a different virus altogether; however, it can carry the genetic messages into human cells and deliver instructions to the cells on how to recognise and defeat the coronavirus. The adenovirus in the vaccine will not replicate in the human body and cause a viral infection.
A single dose of the Johnson & Johnson vaccine is sufficient to immunise a person while other COVID-19 vaccines offer a two-shot immunisation. The vaccine can be kept under refrigerator temperature for months which makes it convenient for transportation and storage in remote and rural areas where it's challenging to bring mRNA vaccines that require freezing temperature. The vaccine is estimated to remain stable for two years at -20°C, and a maximum of 4.5 months at routine refrigeration temperatures of 2° to 8°C, according to the manufacturer [3].

Credit: Freepik
Efficacy of Johnson & Johnson Vaccine
Janssen Ad26.CoV2.S was found to have an efficacy of 85.4 per cent against severe disease and 93.1 per cent against hospitalisation 28 days after vaccination. In clinical trials a single dose of Janssen Ad26.COV2.S was found to have an efficacy of 66.9 per cent against moderate and severe SARS-CoV-2 infection.
Johnson & Johnson's COVID-19 vaccine showed a 72 per cent efficacy rate in a study conducted in the United States. While in South Africa it was found to be 64 per cent. Against severe forms of Covid-19, the vaccine showed 86 per cent efficacy in the US and 82 per cent in South Africa [4].
As per a study published in The New England Journal of Medicine, Johnson & Johnson's vaccine showed an overall efficacy of 66.3 per cent in mild to moderate COVID-19 cases, and an overall 76.3 per cent efficacy in the cases of Severe to critical COVID-19 [5].
Side effects of Johnson & Johnson vaccine
The WHO Strategic Advisory Group of Experts (SAGE) on Immunisation after thoroughly assessing the data on quality, safety and efficacy of the vaccine recommended its use for "people aged 18 and above [6]."
The U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) also reviewed Johnson & Johnson's vaccine and found it to be safe for use.
The vaccine may have some typical side effects associated with all the COCID-19 vaccines: redness, swelling, and pain at the injection site, fever, fatigue, headache, chills, nausea, vomiting, diarrhoea, muscle pain, and/or joint pain [7].
On April 13, 2021, the FDA and CDC called for a pause in the use of the Johnson & Johnson COVID-19 vaccine following a report of a "rare and severe" blood clot in six participants.
However, the pause was lifted a week later after following a safety review [8].
European Medicines Agency (EMA) added Guillain-Barré syndrome, a rare nerve-degeneration disorder, as a possible rare side effect of Johnson & Johnson's Covid-19 vaccine [9].
Is Johnson & Johnson vaccine effective against the Delta variant?
According to an article published on Johnson & Johnson's official website, their single-shot COVID-19 vaccine generated "strong, persistent activity" against the Delta variant [10].
According to a trial, called Sisonke, the single-dose shot showed a 71 per cent efficacy against hospitalisation and as much as 96 per cent efficacy against death [11].
Disclaimer: The information provided in this article is for general informational and educational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or a qualified healthcare provider with any questions you may have regarding a medical condition.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications
