Latest Updates
-
 World Malaria Day 2026: Date, History, Significance, and Why It Matters
World Malaria Day 2026: Date, History, Significance, and Why It Matters -
 Bakery Style Soft Texture Banana Cake Recipe
Bakery Style Soft Texture Banana Cake Recipe -
 Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains
Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains -
 Rich Mughlai Special Chicken Korma Recipe
Rich Mughlai Special Chicken Korma Recipe -
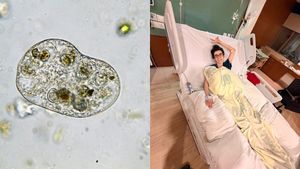 A Hidden Foodborne Infection: What You Should Know About Cyclospora
A Hidden Foodborne Infection: What You Should Know About Cyclospora -
 Melt-in-Mouth Sweet Mysore Pak Recipe: A Classic Indian Delight
Melt-in-Mouth Sweet Mysore Pak Recipe: A Classic Indian Delight -
 Between Meetings and Meals: Why American Pecans Are the Ideal Midday Snack
Between Meetings and Meals: Why American Pecans Are the Ideal Midday Snack -
 Skincare Hacks 101: 7 Summer Hacks That Actually Work in 40°C Heat
Skincare Hacks 101: 7 Summer Hacks That Actually Work in 40°C Heat -
 8 Workouts That Should Be a Part of Everyone’s Lifestyle for Longevity
8 Workouts That Should Be a Part of Everyone’s Lifestyle for Longevity -
 Simple Everyday Dal Recipe: Your Go-To Chana Dal
Simple Everyday Dal Recipe: Your Go-To Chana Dal
Corbevax Will Be Allowed As A Booster For Those Fully Vaccinated With Covishield Or Covaxin
Allowing Biological E's Corbevax as a booster for those fully vaccinated against COVID-19 with either Covishield or Covaxin is likely to be considered by the NTAGI, which is scheduled to meet on Tuesday, sources said.
The Drugs Controller General of India (DCGI) on June 4 approved Corbevax as a precaution dose for those aged 18 and above. India's first indigenously-developed RBD protein subunit vaccine, Corbevax, is currently being used to inoculate children in the age group of 12 to 14 years.
"The National Technical Advisory Group on Immunisation (NTAGI) will discuss allowing Corbevax, which has been cleared by the DCGI, as a booster for those fully vaccinated with Covishield or Covaxin," a source told PTI.
The government advisory panel is also expected to review the trial data of the country's first quadrivalent human papillomavirus (qHPV) vaccine against cervical cancer, developed by the Serum Institute of India.
A separate HPV working group of the NTAGI had, on June 8, examined the vaccine's clinical trial data and usefulness for inclusion in the national immunisation programme, sources had said.
The recommendation of the Standing Technical Sub-Committee (STSC) of the NTAGI on reducing the gap between the second dose and the precaution dose of Covid vaccines from nine to six months may also be ratified at the meeting to be chaired by the Union health secretary. PTI PLB RC
Disclaimer: The information provided in this article is for general informational and educational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or a qualified healthcare provider with any questions you may have regarding a medical condition.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications
