Latest Updates
-
 World Malaria Day 2026: Date, History, Significance, and Why It Matters
World Malaria Day 2026: Date, History, Significance, and Why It Matters -
 Bakery Style Soft Texture Banana Cake Recipe
Bakery Style Soft Texture Banana Cake Recipe -
 Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains
Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains -
 Rich Mughlai Special Chicken Korma Recipe
Rich Mughlai Special Chicken Korma Recipe -
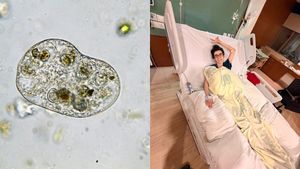 A Hidden Foodborne Infection: What You Should Know About Cyclospora
A Hidden Foodborne Infection: What You Should Know About Cyclospora -
 Melt-in-Mouth Sweet Mysore Pak Recipe: A Classic Indian Delight
Melt-in-Mouth Sweet Mysore Pak Recipe: A Classic Indian Delight -
 Between Meetings and Meals: Why American Pecans Are the Ideal Midday Snack
Between Meetings and Meals: Why American Pecans Are the Ideal Midday Snack -
 Skincare Hacks 101: 7 Summer Hacks That Actually Work in 40°C Heat
Skincare Hacks 101: 7 Summer Hacks That Actually Work in 40°C Heat -
 8 Workouts That Should Be a Part of Everyone’s Lifestyle for Longevity
8 Workouts That Should Be a Part of Everyone’s Lifestyle for Longevity -
 Simple Everyday Dal Recipe: Your Go-To Chana Dal
Simple Everyday Dal Recipe: Your Go-To Chana Dal
Biological E Hopes To Roll Out COVID-19 Vaccine Corbevax By November-End
Biological E. Limited (BE) is expecting its COVID-19 vaccine Corbevax to be rolled out by the end of November even as the city-based firm is getting ready with 100 million doses for the launch, Mahima Datla, Managing Director, BE said on Monday.

Speaking to reporters on the sidelines of signing a financing agreement with US International Development Finance NSE 6.68 % Corporation (DFC), she said currently the manufactured doses are being sent to Central Drugs Laboratory (CDL), Kasauli in Himachal Pradesh for regulatory testing.
"Corbevax is in phase 3 trials. We are likely to complete all the studies by end of November which is when we are expecting licence (from the drug regulator). The licence for children should follow one month later. The study in children is underway as well," she said.
Biological E's anti-coronavirus shot, Corbevax, is a RBD protein sub-unit vaccine and is currently undergoing phase 2/3 clinical trials on adults.
The Phase 3 trials are going to be a study in comparison with the already licensed vaccines, though the study in children is not a comparative study, she further said.
"We are submitting doses to Kasauli for release. So it is my hope that at the day of launch, it has always been our vision to have close to ten crore doses on the day. By the end of November this may happen as soon as we get the license," she said.
On the COVID-19 vaccine manufacturing capacities, she said currently BE has the capacity to make one billion doses of Corbevax per annum, and 600 million doses of Johnson & Johnson's jab.
Biological E Ltd had earlier said it entered into an agreement with Janssen Pharmaceutica NV, part of pharma major Johnson & Johnson, for creation and enhancement of production capabilities to manufacture the latter's COVID-19 vaccine.
Meanwhile, DFC's Chief Operating Officer David Marchick and Mahima Datla today unveiled the expansion of Biological E.'s vaccine manufacturing facility here and finalised a U.S. government financing arrangement formalising USD 50 million to expand the company's capacity to produce COVID-19 vaccines.
"DFC's partnership with Biological E will support capacity for production of more than one billion vaccine doses by the end of 2022 for India and for developing countries around the world," Marchick said in a press release.
"We are pleased with the financial support from the U.S. government, especially DFC, which was announced at the Quad Summit in March 2021. This investment will not only help us augment our capacity to produce more COVID-19 vaccines, but also help the global community that has been relentlessly fighting against the spread of the COVID-19 pandemic," Mahima Datla said.
Disclaimer: The information provided in this article is for general informational and educational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or a qualified healthcare provider with any questions you may have regarding a medical condition.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications
