Latest Updates
-
 Horoscope for Today April 29, 2026 - Calm Focus & Steady Progress
Horoscope for Today April 29, 2026 - Calm Focus & Steady Progress -
 Maharashtra Day 2026: Why The State Of Bombay Split Into Two And How The Day Is Marked With Celebrations
Maharashtra Day 2026: Why The State Of Bombay Split Into Two And How The Day Is Marked With Celebrations -
 Vegetable Kurma Recipe: Experience South Indian Authentic Taste
Vegetable Kurma Recipe: Experience South Indian Authentic Taste -
 Home Cooling Tips Without An Air Conditioner This Summer – 6 Decor Solutions That Do The Trick!
Home Cooling Tips Without An Air Conditioner This Summer – 6 Decor Solutions That Do The Trick! -
 Sweet Spicy Street Style Honey Chilli Potato Recipe
Sweet Spicy Street Style Honey Chilli Potato Recipe -
 Beauty Hacks 101: 5 Easy Ways To Use Mango For Quick Summer Skin Fixes At Home
Beauty Hacks 101: 5 Easy Ways To Use Mango For Quick Summer Skin Fixes At Home -
 Two Teachers Die On Census Duty In Odisha, Heatstroke Suspected: Why Summers Are Getting Harder To Survive
Two Teachers Die On Census Duty In Odisha, Heatstroke Suspected: Why Summers Are Getting Harder To Survive -
 Veg Fried Rice Recipe: Master the Wok Toss Restaurant Trick
Veg Fried Rice Recipe: Master the Wok Toss Restaurant Trick -
 Samantha Ruth Prabhu Birthday Special: 7 Outfits That Redefined ‘The Family Man 2’ Star’s Power Dressing
Samantha Ruth Prabhu Birthday Special: 7 Outfits That Redefined ‘The Family Man 2’ Star’s Power Dressing -
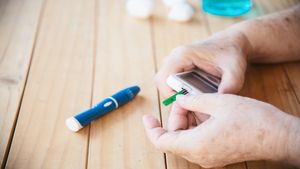 Hidden Signs Of Insulin Resistance: What Your Body Is Trying To Tell You
Hidden Signs Of Insulin Resistance: What Your Body Is Trying To Tell You
AIIMS Delhi To Start Booster Dose Trial Of Intranasal Covid Vaccine From March 11
The AIIMS will start the booster dose trial of Bharat Biotech's intranasal COVID-19 vaccine from Friday. The booster dose will be given to those who have received both the doses of either Covaxin or Covishield at least five months ago but not more than seven months back, Dr Sanjay Rai, Professor at the Centre for Community Medicine at AIIMS, New Delhi told PTI.

India is yet to approve the use of intranasal vaccine against COVID-19, BBV154 developed by the Hyderabad based company.
The Drugs Controller General of India (DCGI) in January had given permission to Bharat Biotech to conduct a phase-3 randomized, multi-centric study to evaluate the immunogenicity and safety of the booster dose in participants previously vaccinated with Covishield or Covaxin.
"We got the ethical approval for initiating the booster dose trial on Wednesday. Registration for the participants will start from March 10 via email --HYPERLINK "mailto:[email protected]"[email protected] --- and WhatsApp (7428847499) for those who received both the doses of either Covaxin or Covishield more than 5 months but less than 7 months ago," Dr Rai said.
AIIMS, New Delhi is one of the five sites where the booster trial for the nasal vaccine will be conducted.
BBV154 is an intranasal replication-deficient chimpanzee adenovirus SARS-CoV-2 vectored vaccine.
Disclaimer: The information provided in this article is for general informational and educational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or a qualified healthcare provider with any questions you may have regarding a medical condition.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications
