Latest Updates
-
 Hair Hacks 101: How To Make Your Hair Smell Fresh Without Washing It Daily
Hair Hacks 101: How To Make Your Hair Smell Fresh Without Washing It Daily -
 New LPG Rules From May 1, 2026: OTP Delivery, Price Changes And Booking Limits You Must Know
New LPG Rules From May 1, 2026: OTP Delivery, Price Changes And Booking Limits You Must Know -
 Why Your Gut Feels Different During Your Period: Expert Explains
Why Your Gut Feels Different During Your Period: Expert Explains -
 Buddha Purnima 2026: When Is It? Significance, Rituals, And How To Observe
Buddha Purnima 2026: When Is It? Significance, Rituals, And How To Observe -
 Classic Home Style Curry: Mutter Paneer Recipe
Classic Home Style Curry: Mutter Paneer Recipe -
 Raja Ravi Varma's 178th Birth Anniversary: 6 South Indian Actresses Who Brought His Paintings To Life
Raja Ravi Varma's 178th Birth Anniversary: 6 South Indian Actresses Who Brought His Paintings To Life -
 Fire Breaks Out At Ghaziabad Society — Here's How To Stay Alive In A High-Rise Emergency
Fire Breaks Out At Ghaziabad Society — Here's How To Stay Alive In A High-Rise Emergency -
 International Dance Day 2026: Why Dance Workouts Are Becoming The Most Engaging Fitness Trend
International Dance Day 2026: Why Dance Workouts Are Becoming The Most Engaging Fitness Trend -
 Light Creamy Morning Bowl Mushroom Soup Recipe
Light Creamy Morning Bowl Mushroom Soup Recipe -
 Horoscope for Today April 29, 2026 - Calm Focus & Steady Progress
Horoscope for Today April 29, 2026 - Calm Focus & Steady Progress
Each Dose Of Covishield, Covaxin Likely To Be Capped At Rs 275 After Getting Regular Market Approval
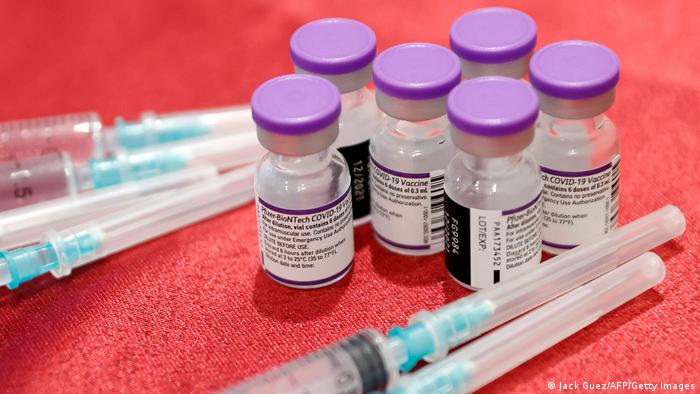
The price of Covishield and Covaxin, the COVID-19 vaccines which are expected to soon get regular market approval from India's drug regulator, is likely to be capped at Rs 275 per dose plus an additional service charge of Rs 150, official sources said. According to them, the National Pharmaceutical Pricing Authority (NPPA) has been directed to start working towards capping the price to make the vaccines affordable.
As of now, Covaxin is priced at Rs 1,200 per dose while Covishield costs Rs 780 in private facilities. The prices include Rs 150 service charge. Both the vaccines are only authorised for emergency use in the coAn Subject Expert Committee on COVID-19 of the Central Drugs Standard Control Organisation on January 19 recommended granting regular market approval to COVID vaccines Covishield and Covaxin for use in the adult population subject to certain conditions.
"The NPPA has been asked to work towards capping the price of the vaccines. The price is likely to be capped at Rs 275 per dose along with an additional service charge of Rs 150," an official source said.
Prakash Kumar Singh, the director (government and regulatory affairs) at Serum Institute of India, had submitted an application to the Drugs Controller General of India on October 25 seeking regular market approval for its Covishiled vaccine.
A couple of weeks ago, V Krishna Mohan, the whole-time director at Bharat Biotech, submitted complete information on the chemistry, manufacturing and controls, along with the pre-clinical and clinical data while seeking regular market authorisation for Covaxin.
Covaxin and Covishield were granted Emergency Use Authorisation (EUA) on January 3 last year.
COVID vaccine - Photo by DW News / Getty Images.
Disclaimer: The information provided in this article is for general informational and educational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or a qualified healthcare provider with any questions you may have regarding a medical condition.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications
