Latest Updates
-
 Besan Ladoo Recipe: Your Guide to Homemade Sweet Perfection
Besan Ladoo Recipe: Your Guide to Homemade Sweet Perfection -
 Horoscope for Today April 21, 2026 - Steady Progress & Calm Focus
Horoscope for Today April 21, 2026 - Steady Progress & Calm Focus -
 Street Style Protein Dish: Soya Chaap Recipe
Street Style Protein Dish: Soya Chaap Recipe -
 How Middle-Class Families in India Can Choose the Right Health Insurance
How Middle-Class Families in India Can Choose the Right Health Insurance -
 Pizza Dough Recipe: Your Perfect Base Every Time
Pizza Dough Recipe: Your Perfect Base Every Time -
 DIY Skincare in Summer: What Actually Helps vs What Can Irritate Your Skin
DIY Skincare in Summer: What Actually Helps vs What Can Irritate Your Skin -
 The Biggest Skincare Mistakes Teens Make And How to Fix Them, Expert Insights
The Biggest Skincare Mistakes Teens Make And How to Fix Them, Expert Insights -
 “Online Rape Academy” Exposed: Inside the Disturbing Network Behind the Viral Claim
“Online Rape Academy” Exposed: Inside the Disturbing Network Behind the Viral Claim -
 ₹44,800 Ralph Lauren Bandhani Skirt Ignites Cultural Appropriation Debate
₹44,800 Ralph Lauren Bandhani Skirt Ignites Cultural Appropriation Debate -
 Litti Chokha Recipe: A Taste of Bihar Traditional Delight
Litti Chokha Recipe: A Taste of Bihar Traditional Delight
COVID-19 Vaccine: Government Panel Recommends EUA For Corbevax For Kids Between 5-11 Years Age Groups
An expert panel of India's central drug authority has recommended granting emergency use authorisation for Biological E's COVID-19 vaccine Corbevax for children in the five to 11 years age group, official sources said on Thursday.
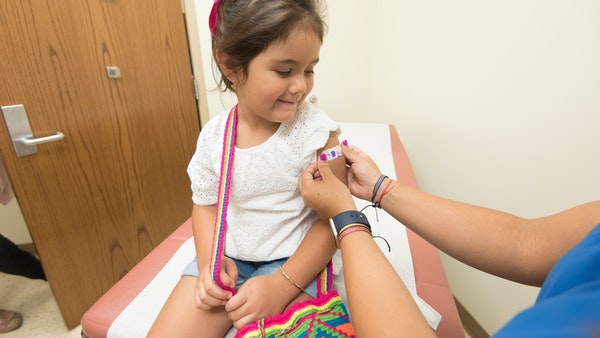
The Subject Expert Committee on COVID-19 of the CDSCO has, however, sought more data from Bharat Biotech to review its emergency use authorisation (EUA) application for use of Covaxin among those aged between two and 11 years, they said.
The Drugs Controller General of India (DCGI) had approved Covovax for restricted use in emergency situations in adults on 28 December last year and in the 12 to 17 years age group, subject to certain conditions, on 9 March this year.
Biological E's Corbevax is being used to inoculate children against COVID-19 in the age group of 12 to 14 years. Covaxin has been granted Emergency Use Listing (EUL) by the DCGI for the age group of 12 to 18 years on 24 December 2021.

"The Subject Expert Committee on COVID-19 of the CDSCO which deliberated on Biological E's EUA application has recommended granting emergency use authorisation for use of Corbevax in the age group of five to 11 years," a source said.
India began inoculating children aged 12-14 on 16 March.
The countrywide vaccination drive was rolled out on 16 January last year with healthcare workers getting inoculated in the first phase. Vaccination of frontline workers started on 2 February last year.
The next phase of COVID-19 vaccination commenced on 1 March last year for people over 60 years of age and those aged 45 and above with specified co-morbid conditions.
India launched vaccination for all people aged more than 45 years on 1 April last year. The government then decided to expand its vaccination drive by allowing everyone above 18 years of age to be inoculated against the viral disease from 1 May last year.
The next phase of vaccination commenced on 3 January for adolescents in the age group of 15-18 years.
India began administering precaution doses of vaccines to healthcare and frontline workers and those aged 60 and above with comorbidities on 10 January.
Precaution dose of COVID-19 vaccines to all aged above 18 years was allowed at private vaccination centres from 10 April.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications
