Latest Updates
-
 The Creamy Side Dish Trick: Perfect Mashed Potato Recipe
The Creamy Side Dish Trick: Perfect Mashed Potato Recipe -
 Uranus In Gemini Transit Returns For The First Time Since The 1940s, Starts A 7-Year Shift Across Zodiac Signs
Uranus In Gemini Transit Returns For The First Time Since The 1940s, Starts A 7-Year Shift Across Zodiac Signs -
 Sita Navami 2026: Puja Muhurat, Vrat Vidhi And Spiritual Benefits For Stronger Relationships
Sita Navami 2026: Puja Muhurat, Vrat Vidhi And Spiritual Benefits For Stronger Relationships -
 World Malaria Day 2026: Date, History, Significance, and Why It Matters
World Malaria Day 2026: Date, History, Significance, and Why It Matters -
 Bakery Style Soft Texture Banana Cake Recipe
Bakery Style Soft Texture Banana Cake Recipe -
 Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains
Horoscope for Today April 25, 2026 - Steady Steps, Clear Focus, Practical Gains -
 Rich Mughlai Special Chicken Korma Recipe
Rich Mughlai Special Chicken Korma Recipe -
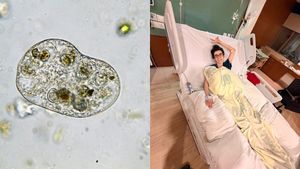 A Hidden Foodborne Infection: What You Should Know About Cyclospora
A Hidden Foodborne Infection: What You Should Know About Cyclospora -
 Melt-in-Mouth Sweet Mysore Pak Recipe: A Classic Indian Delight
Melt-in-Mouth Sweet Mysore Pak Recipe: A Classic Indian Delight -
 Between Meetings and Meals: Why American Pecans Are the Ideal Midday Snack
Between Meetings and Meals: Why American Pecans Are the Ideal Midday Snack
India-US Healthcare Collaboration In Spotlight As India Approves Two More Covid-19 Vaccines
India-US collaboration in the health sector is in the spotlight as the Central Drug Authority this week approved two more Covid vaccines Corbevax and Covovax and antiviral drug Molnupiravir for use in India.

Taranjit Singh Sandhu, India's Ambassador to the US, in a tweet described it as a model of India-US healthcare collaboration. "Models of what India-United States healthcare collaboration can achieve for global good!" Sandhu said.
Indian companies working with Texas Children's, Baylor College of Medicine's National School of Tropical Medicine; Dr Peter Hotez, Prof and Dean of the National School of Tropical Medicine at Baylor and Co-Director of the Texas Children's Hospital Center for Vaccine Development; NovaVax; Merck and Ridgeback Bio, tweeted the Indian diplomat.
During his visit to Houston in October, Sandhu had met Professor Hotez and held discussions on this issue. During his visit to Houston in October, Sandhu had met Professor Hotez and held discussions on this issue.
In June this year, the ambassador visited the Novavax facility in Maryland. He also spoke to the CEO of Sanisure Thomas Hook. Sanisure supplies components for the SII - Novavax collaboration.
These have been part of a continuous outreach that the ambassador had with vaccine manufacturers and pharma companies. Corbevax, a protein sub-unit COVID-19 vaccine, whose technology was created and engineered in collaboration with Texas Children's Hospital and Baylor College of Medicine has received Emergency Use Authorization (EUA) approval from the Drugs Controller General of India (DCGI).
The vaccine is being launched in India with other underserved countries to follow, Texas Children's Hospital said in a statement. "This announcement is an important first step in vaccinating the world and halting the pandemic. Our vaccine technology offers a path to address an unfolding humanitarian crisis, namely the vulnerability the low- and middle-income countries face against the delta variant, Hotez said.
"Widespread and global vaccination with our Texas Children's-Baylor-BE vaccine would also forestall the emergence of new variants. We have previously missed that opportunity for the alpha and delta variants. Now is our chance to prevent a new global wave from what might follow, he said.
Dubbed the world's COVID-19 vaccine, it uses a traditional recombinant protein-based technology that will enable its production at large scales making it widely accessible to inoculate the global population, the statement said.
Corbevax after completing two Phase III clinical trials involving more than 3,000 subjects was found to be safe, well-tolerated and immunogenic.
It demonstrated superior immune response in comparison with the Covishield vaccine when assessed for Neutralising Antibody (nAb) Geometric Mean Titers (GMT) against the Ancestral-Wuhan strain and the globally dominant Delta variant.
Corbevax vaccination also generated a significant Th1 skewed cellular immune response.
Disclaimer: The information provided in this article is for general informational and educational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or a qualified healthcare provider with any questions you may have regarding a medical condition.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications
