Latest Updates
-
 Tangy Italian Classic Red Sauce Pasta Recipe
Tangy Italian Classic Red Sauce Pasta Recipe -
 Who Was Divyanka Sirohi? Haryanvi Actress Passes Away At 30 After Reported Heart Attack, Old Post Goes Viral
Who Was Divyanka Sirohi? Haryanvi Actress Passes Away At 30 After Reported Heart Attack, Old Post Goes Viral -
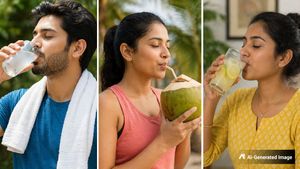 ORS vs Coconut Water vs Nimbu Pani: Which Is Really Better For Rehydration?
ORS vs Coconut Water vs Nimbu Pani: Which Is Really Better For Rehydration? -
 Summer Diet vs All-Season Diet: What Really Changes When the Heat Kicks In?
Summer Diet vs All-Season Diet: What Really Changes When the Heat Kicks In? -
 Puran Poli Recipe: Your Guide to Maharashtrian Festive Sweet Delights
Puran Poli Recipe: Your Guide to Maharashtrian Festive Sweet Delights -
 India’s Waste Policy Will Fail Without Low-Value Plastic Recovery: The Overlooked 70% Packaging Growing Crisis
India’s Waste Policy Will Fail Without Low-Value Plastic Recovery: The Overlooked 70% Packaging Growing Crisis -
 Body Acne in Summer: Why It Happens and How to Deal With It
Body Acne in Summer: Why It Happens and How to Deal With It -
 70% People Use AI Astrology For Love Problems, Not Career: Check What This Report Says
70% People Use AI Astrology For Love Problems, Not Career: Check What This Report Says -
 7 Must-Have Items to Carry Daily to Beat the Summer Heat
7 Must-Have Items to Carry Daily to Beat the Summer Heat -
 How Overall Health Impacts Hair Growth
How Overall Health Impacts Hair Growth
Sun Pharma Gets DCGI Nod To Market Molnupiravir For COVID-19 Treatment
Sun Pharma on Tuesday said its subsidiary has received emergency use authorisation (EUA) from the Drugs Controller General of India (DCGI) to manufacture and market a generic version of MSD and Ridgeback's antiviral drug molnupiravir under the brand name Molxvir in India.
Earlier this year, the Mumbai-based drug major had signed a non-exclusive voluntary licensing agreement with MSD to manufacture and supply a generic version of molnupiravir in over 100 low and middle-income countries (LMICs) including India.
The DCGI has approved molnupiravir for the treatment of adult patients with COVID-19 and who have a high risk of progression of the disease including hospitalisation or death.
Molnupiravir is an important addition to the portfolio of oral therapies available for treating Covid-19 patients, Sun Pharmaceutical Industries CEO (India Business) Kirti Ganorkar said in a statement.
"In line with our consistent efforts to accelerate access to new drugs for Covid-19 treatment, we will make Molxvir available to patients at an affordable price. We are also in the process of launching a toll-free helpline to ensure the availability of Molxvir to doctors and patients across India. Our endeavour is to make the product available in a week's time," he added.
The recommended dose of the drug is 800 mg twice a day for five days. The duration of treatment of molnupiravir is much shorter compared to other therapies which is a significant advantage as it reduces the pill burden and enhances compliance.
Drug major Cipla and Torrent Pharmaceuticals on Tuesday also said they plan to launch the antiviral drug Molnupiravir for the treatment of adult patients with COVID-19, who have a high risk of progression of the disease including hospitalization or death, under the brand name Cipmolnu.
Dr Reddy's Laboratories to Introduce Molnupiravir to Treat Covid-19 Shortly
Amid Concerns Over Omicron, US Health Panel Says 'Merck Pill Can Prevent Grave Covid Infections'
Earlier this year, Torrent Pharma inked a non-exclusive voluntary licensing pact with MSD for manufacturing, distribution and marketing of Molnupiravir in more than 100 low-and middle-income markets, including India.
Molnupiravir has been developed by MSD and Ridgeback Biotherapeutics. It has been approved by the US Food and Drug Administration (FDA) and the UK Medicines and Healthcare products Regulatory Agency (MHRA) for Emergency Use Authorisation (EUA).
Disclaimer: The information provided in this article is for general informational and educational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or a qualified healthcare provider with any questions you may have regarding a medical condition.



 Click it and Unblock the Notifications
Click it and Unblock the Notifications












