Just In
- 1 hr ago

- 2 hrs ago

- 6 hrs ago

- 10 hrs ago

Don't Miss
- Technology
 OnePlus Ace 3 Pro Leak Hints at New Design; Expected Launch, Specifications We Know So Far
OnePlus Ace 3 Pro Leak Hints at New Design; Expected Launch, Specifications We Know So Far - News
 Euro Zone Economy Faces Risks Without Interest Rate Cuts: ECB Member Warns
Euro Zone Economy Faces Risks Without Interest Rate Cuts: ECB Member Warns - Sports
 IPL 2024: More pressure on Hardik Pandya as Dale Steyn suggests MI captain to step up and help Jasprit Bumrah
IPL 2024: More pressure on Hardik Pandya as Dale Steyn suggests MI captain to step up and help Jasprit Bumrah - Movies
 Coachella: AP Dhillon's Tribute To Sidhu Moosewala Goes Viral Amid Guitar Controversy, Fans REACT To VIDEO
Coachella: AP Dhillon's Tribute To Sidhu Moosewala Goes Viral Amid Guitar Controversy, Fans REACT To VIDEO - Finance
 Rs 14/Share Dividend: Stockbroking Co To Consider Bonus Share, Shares Up 126% In 180-Days
Rs 14/Share Dividend: Stockbroking Co To Consider Bonus Share, Shares Up 126% In 180-Days - Travel
 Journey From Delhi To Ooty: Top Transport Options And Attractions
Journey From Delhi To Ooty: Top Transport Options And Attractions - Education
 IIIT-Bangalore Introduces PG Diploma In Digital Product Design And Management
IIIT-Bangalore Introduces PG Diploma In Digital Product Design And Management - Automobiles
 Jawa Yezdi Expands Mega Service Camps To 32 New Cities, Focusing On Tier-II And Tier-III Regions
Jawa Yezdi Expands Mega Service Camps To 32 New Cities, Focusing On Tier-II And Tier-III Regions
Each Dose Of Covishield, Covaxin Likely To Be Capped At Rs 275 After Getting Regular Market Approval
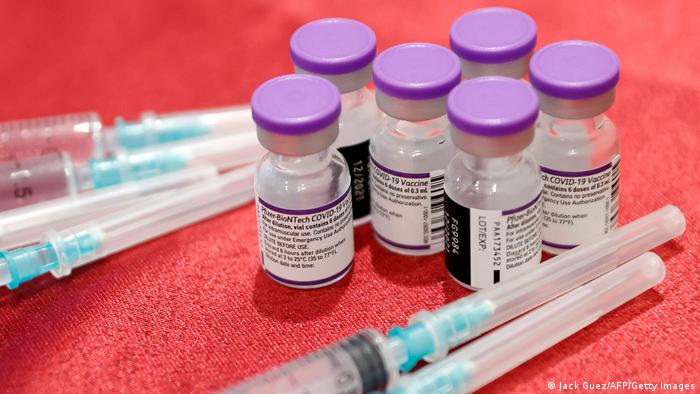
The price of Covishield and Covaxin, the COVID-19 vaccines which are expected to soon get regular market approval from India's drug regulator, is likely to be capped at Rs 275 per dose plus an additional service charge of Rs 150, official sources said. According to them, the National Pharmaceutical Pricing Authority (NPPA) has been directed to start working towards capping the price to make the vaccines affordable.
As of now, Covaxin is priced at Rs 1,200 per dose while Covishield costs Rs 780 in private facilities. The prices include Rs 150 service charge. Both the vaccines are only authorised for emergency use in the coAn Subject Expert Committee on COVID-19 of the Central Drugs Standard Control Organisation on January 19 recommended granting regular market approval to COVID vaccines Covishield and Covaxin for use in the adult population subject to certain conditions.
"The NPPA has been asked to work towards capping the price of the vaccines. The price is likely to be capped at Rs 275 per dose along with an additional service charge of Rs 150," an official source said.
Prakash Kumar Singh, the director (government and regulatory affairs) at Serum Institute of India, had submitted an application to the Drugs Controller General of India on October 25 seeking regular market approval for its Covishiled vaccine.
A couple of weeks ago, V Krishna Mohan, the whole-time director at Bharat Biotech, submitted complete information on the chemistry, manufacturing and controls, along with the pre-clinical and clinical data while seeking regular market authorisation for Covaxin.
Covaxin and Covishield were granted Emergency Use Authorisation (EUA) on January 3 last year.
COVID vaccine - Photo by DW News / Getty Images.
-
 pregnancy parentingMysterious Pneumonia Outbreak In China: What Are The Long-Term Effects Of Pneumonia In Children?
pregnancy parentingMysterious Pneumonia Outbreak In China: What Are The Long-Term Effects Of Pneumonia In Children? -
 healthCan You Get Back COVID-Related Loss Of Sense Of Smell And Taste (Parosmia)? Looks Like You Can!
healthCan You Get Back COVID-Related Loss Of Sense Of Smell And Taste (Parosmia)? Looks Like You Can! -
 healthDid Covid-10 Vaccination Increase Risk Of Sudden Deaths In Young Adults? ICMR Finds This..
healthDid Covid-10 Vaccination Increase Risk Of Sudden Deaths In Young Adults? ICMR Finds This.. -
 wellnessDelhi Air Pollution, Air Quality Very Poor: Do Covid Masks Help Reduce Pollution Side Effects?
wellnessDelhi Air Pollution, Air Quality Very Poor: Do Covid Masks Help Reduce Pollution Side Effects? -
 healthNobel Prize 2023: Scientists Behind The COVID-19 mRNA Vaccines Wins
healthNobel Prize 2023: Scientists Behind The COVID-19 mRNA Vaccines Wins -
 healthWhat Is Disease X? 20 Times More Deadlier Than Covid 19; Can Cause 50 Million Deaths, Does It Have Vaccine?
healthWhat Is Disease X? 20 Times More Deadlier Than Covid 19; Can Cause 50 Million Deaths, Does It Have Vaccine? -
 healthCOVID-19 Vaccines and Heart Attacks: New Studies Show Different Results; What Are They?
healthCOVID-19 Vaccines and Heart Attacks: New Studies Show Different Results; What Are They? -
 healthDo You Pick Your Nose? Stop! It Can Increase COVID Risk
healthDo You Pick Your Nose? Stop! It Can Increase COVID Risk -
 healthEris Variant: WHO Alarms New Covid Wave; Should India Be Concerned About This Pandemic
healthEris Variant: WHO Alarms New Covid Wave; Should India Be Concerned About This Pandemic -
 healthVitamin D Deficiency Linked With Increased Risk Of Long COVID
healthVitamin D Deficiency Linked With Increased Risk Of Long COVID -
 healthCOVID Surge In India: Do You Need A COVID-19 Booster Shot?
healthCOVID Surge In India: Do You Need A COVID-19 Booster Shot? -
 healthCOVID-19: IMA Cites 3 Reasons Behind Recent Surge In Covid-19 Cases: What Are They?
healthCOVID-19: IMA Cites 3 Reasons Behind Recent Surge In Covid-19 Cases: What Are They?


 Click it and Unblock the Notifications
Click it and Unblock the Notifications



